
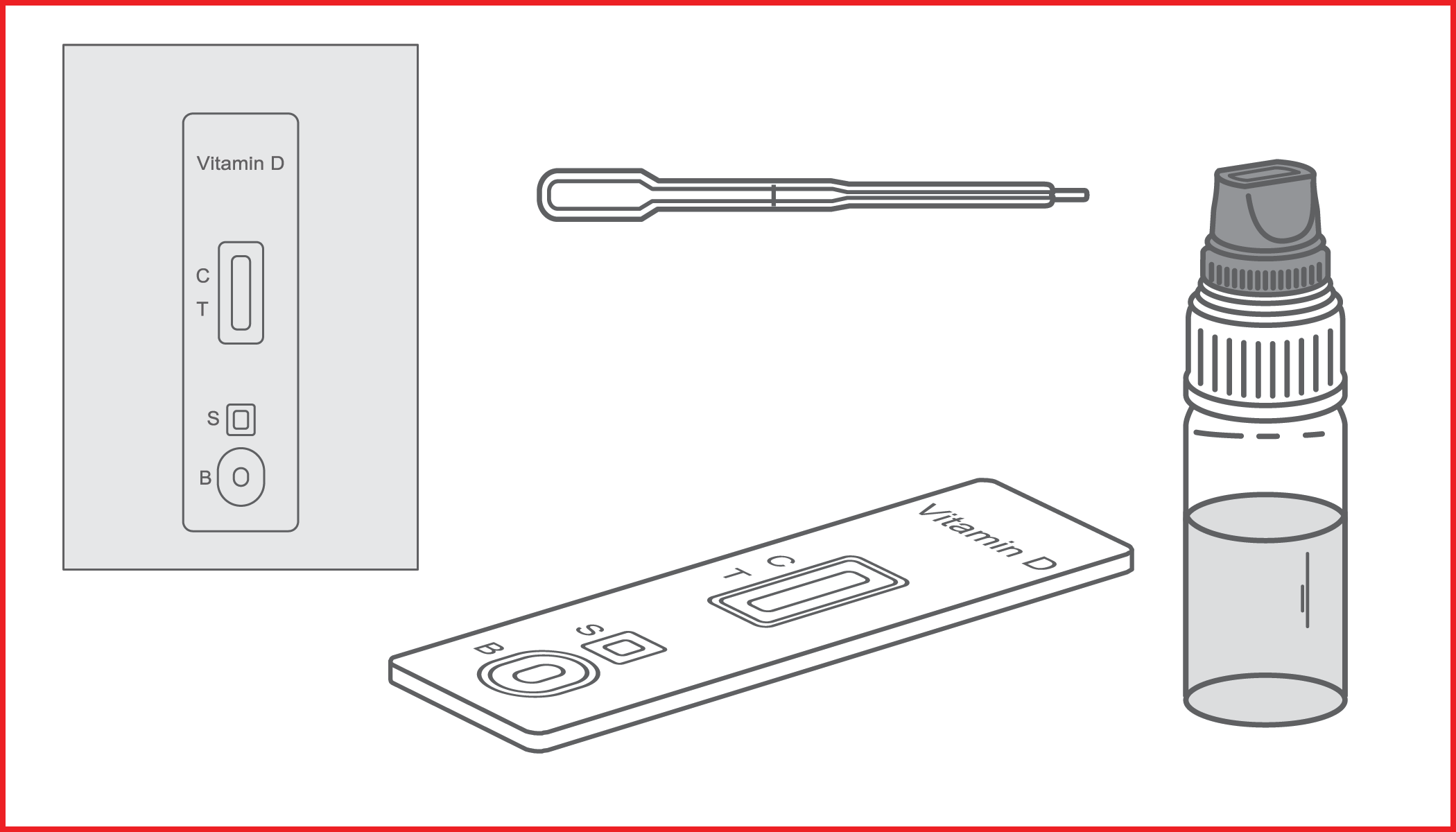
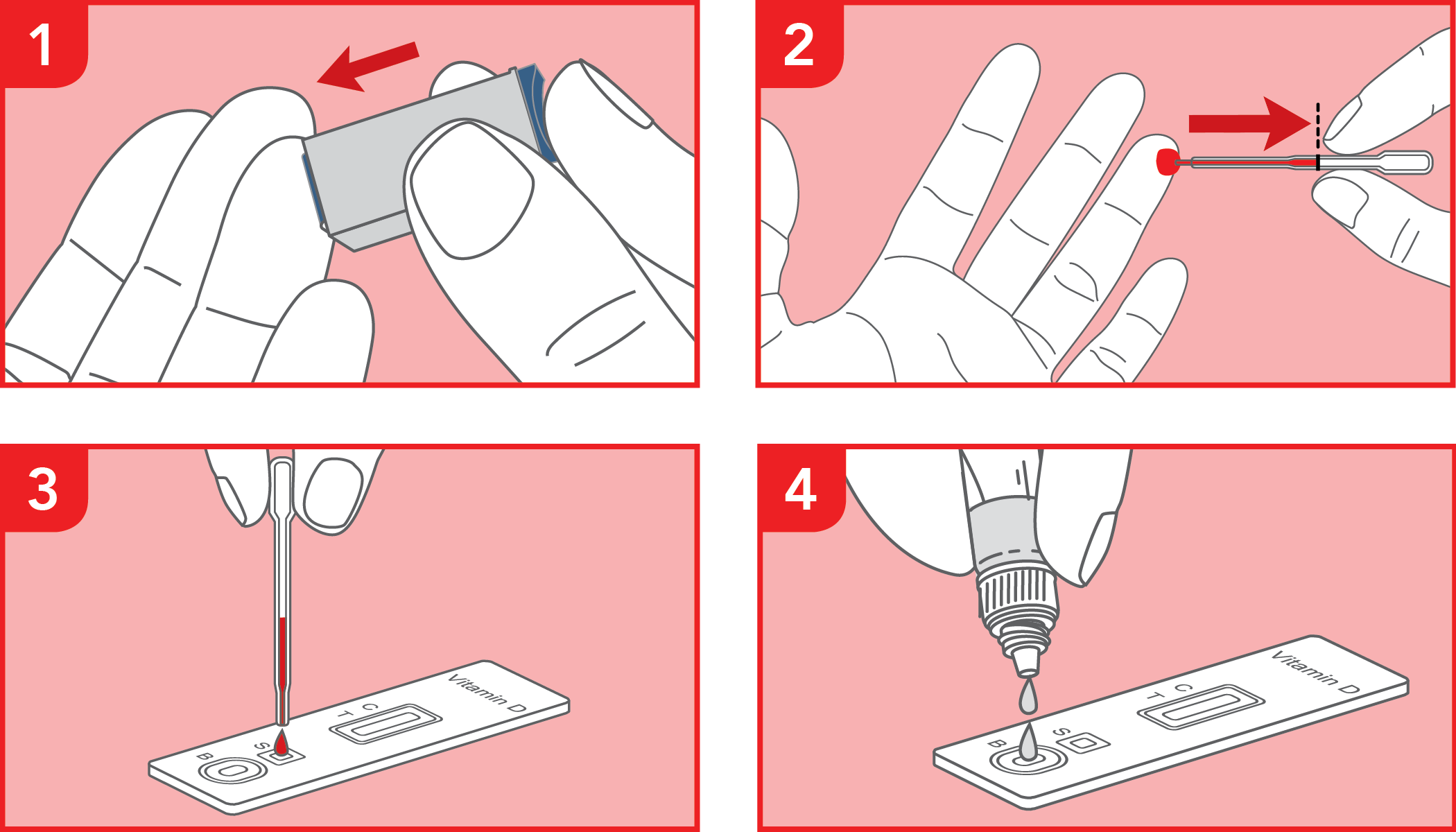

Rapid immunochromatographic test for the semi-quantitative detection of 25-hydroxy vitamin D in human whole blood. IVD for professional use only.

Vitamin D belongs to a group of fat-soluble secosteroids responsible for increasing the intestinal absorption of calcium, iron, magnesium, phosphate and zinc. In humans, total vitamin D refers to the sum of the two main forms of vitamin D, namely vitamin D2 and vitamin D3.
read moreVitamin D is transported to the liver, where it is metabolized to 25-hydroxyvitamin D. The blood test for 25-hydroxyvitamin D is used to determine the concentration of vitamin D in the body. Blood concentration of 25-hydroxyvitamin D (which includes D2 and D3) is considered the best indicator of the level of vitamin D in the body.
Easy to use
Results in 10 minutes
Rapid support to healthcare personnel
VITAMIN D RAPID TEST is a rapid chromatographic immunoassay that offers a preliminary diagnostic result and can be used to screen for vitamin D deficiency, which today is recognized as a worldwide epidemic.
read moreVirtually every cell in our body has vitamin D receptors: this means that they all need a “sufficient” level of vitamin D to function properly. Health risks associated with vitamin D deficiency are much more serious than previously thought. Vitamin D deficiency has been linked to several serious diseases: osteoporosis, osteomalacia, multiple sclerosis, cardiovascular diseases, complications during pregnancy, diabetes, depression, stroke, autoimmune diseases, influenza, various types of cancer, infectious diseases, Alzheimer’s, obesity etc. Therefore, today detecting the level of (25-OH) vitamin D is considered as a “medically necessary screening test”, and maintaining sufficient levels of this vitamin does not only improve bone health, but also health and well-being overall.
VITAMIN D RAPID TEST is an immunoassay based on the principle of competitive protein binding. The 25 (OH) D antigens are pre-fixed on the test line region of the reactive membrane. During the test, the mixture migrates chromatographically along the membrane by capillary action. The 25 (OH) D present in the sample competes with the 25 (OH) D on the test line for limited amounts of anti-25 OH vitamin D antibodies in the conjugate.
| Cut-off di 30 ± 4ng/m | |
| Measuring range | 10-100 ng/mL |
| Accuracy | 94.4% |



Excessive (> 100 ng/mL): no clear lines appear on the test line region (T).
Sufficient (30-100 ng/mL): the intensity of the test line (T) is equal to or less than that of the 30 ng/mL displayed on the color card.
Insufficient (30-10 ng/mL): the intensity of the test line (T) is darker than the 30 ng/mL line and lighter than the 10 ng/mL line displayed on the color card.
Deficit (0-10 ng/mL): the intensity of the test line (T) is equal to or darker than the 10 ng/ mL line displayed on the color card.
1. Holick MF (March 2006). „High prevalence of vitamin D inadequacy and implications for health.“ Mayo Clinic Proceedings. 81 (3): 353–73
2. Eriksen EF, Glerup H (2002). Vitamin D deficiency and aging: implications for general health and osteoporosis. Biogerontology. 3 (1-2): 73-7
3. Grant WB, Holick MF (June 2005). Benefits and requirements of vitamin D for optimal health: a review. Alternative Medicine Review.10 (2): 94-111
4. Moyad MA. Vitamin D: a rapid review. DermatolNurs. 2009, 21:25-30
The test has been carried out correctly when the instructions for use are followed. It includes the reading time and the interpretation of the results shown at the "RESULTS INTERPRETATION" section of the instructions for use.
A colored line will appear at the control region (C) on the test device, showing that the test performed correctly. The absence of the colored line suggests to repeat the test with a new device and a new sample.
The color and intensity of the lines do not affect the interpretation of the result. The test has to be considered positive regardless of the color intensity of the test line (T).
Check product availability with the local representative in your country